Important note: MarinTrust only accept new applications against to the requirements of Version 3 of the Factory Standard and Fishery Assessment. More information about the transition to Version 3.0 can be found via this link.
Application process
The approval process for the MarinTrust Improver Programme is illustrated below (enlarge image):
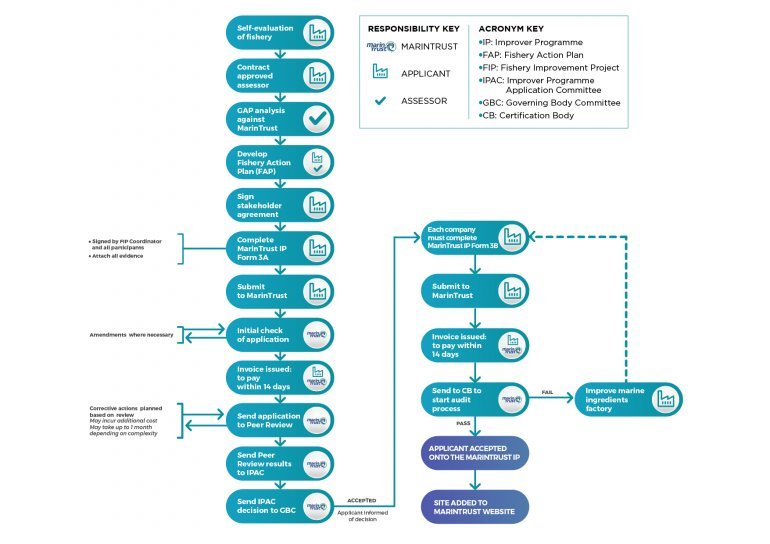
Applicant requirements:
The marine ingredient production facility must be a signed stakeholder in, and be sourcing from, an accepted MarinTrust FIP.
The production facility must pass a MarinTrust audit by a third-party certification body using MarinTrust accepted FIP raw material.
Next steps once the production facility is accepted onto the MarinTrust IP
For the production facility to maintain acceptance on the IP, they must comply with the following:
- The accepted FIP they are sourcing from must comply with the Fishery Action Plan (FAP) and pass annual reporting and peer reviews of this reporting. This is checked each year via the peer review process outlined in the diagrams below.
- The production facility must continue to comply with the MarinTrust audit requirements.
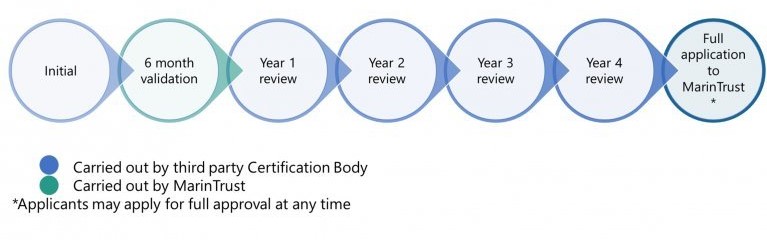
The annual peer review by the CB is to ensure:
- Sufficient evidence provided to demonstrate actions as part of the FAP.
- Identify any areas where the evidence is insufficient.
- Identify any milestones which have not been addressed.
- Make a determination as to whether the FIP has demonstrated appropriate improvements to maintain acceptance on the IP.
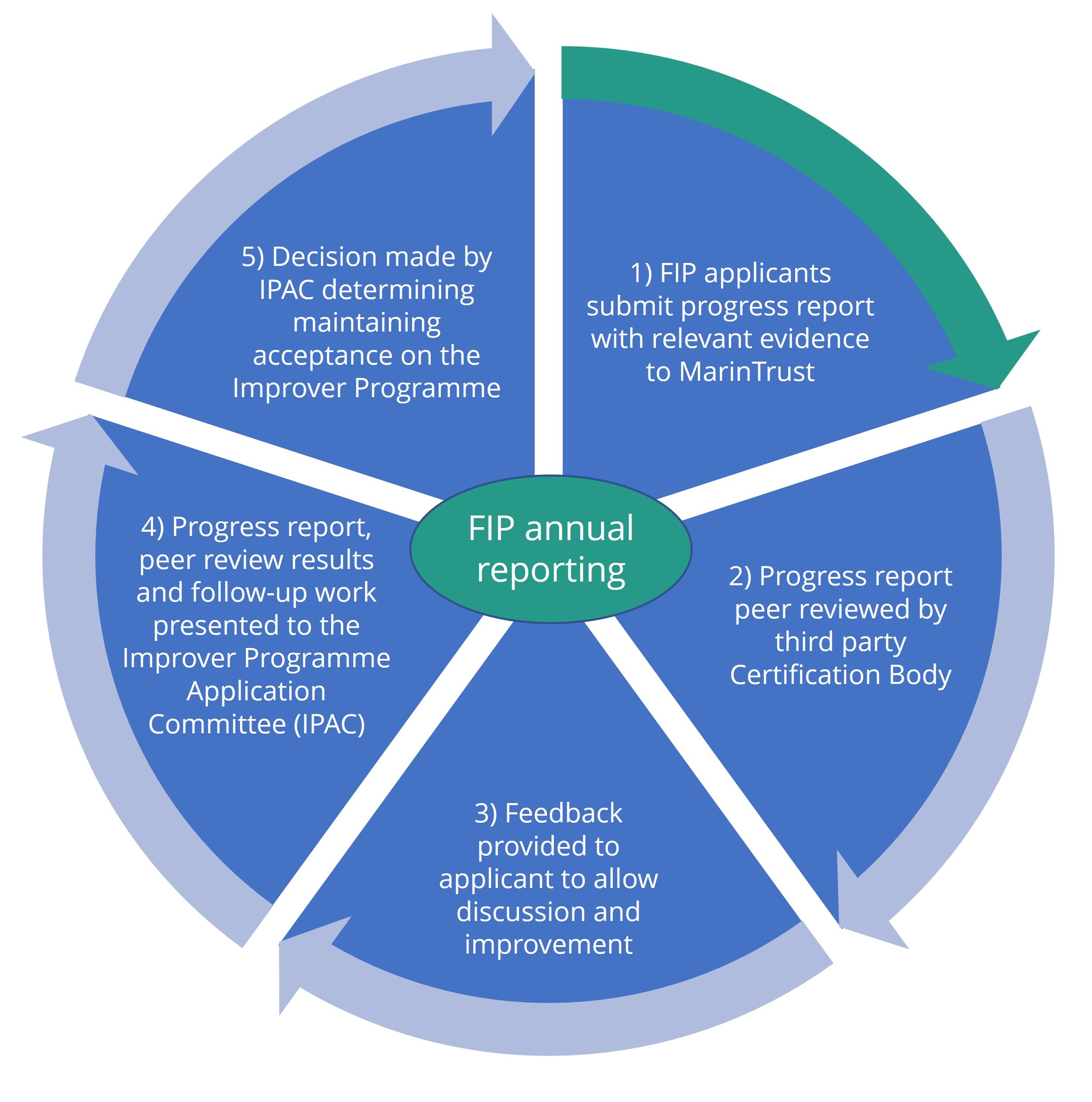
FIP annual reporting process
Claims and benefits
- Read more about the Improver Programme, its benefits and how to make a claim.
Costs
Other questions

